Aiming to Be a Trusted Medical Partner
In Japan, our Medical Representatives (MRs) are responsible for providing, gathering, and relaying information about our pharmaceutical products to healthcare professionals, including doctors and pharmacists. We offer a broad portfolio of medications spanning therapeutic areas such as cardiovascular diseases (e.g., thrombosis and embolism), lifestyle-related diseases (e.g., hypertension and diabetes), central nervous system disorders (e.g., migraine, insomnia, epilepsy), pain, and oncology.
With a well-developed training system, we try to acquire not only information on our own pharmaceuticals, but also a range of knowledge about diseases and pathological conditions. From professional information on the safety and efficacy of drugs that is useful for healthcare professionals who treat patients with various diseases, to information that helps appropriate patients who take our medicines lead healthy and fulfilling lives with their families, we commit ourselves to communicating information that meets the wide-ranging needs of healthcare professionals in an accurate, prompt and thorough manner.
We also ensure strict compliance through ongoing training and thorough recordkeeping, aiming to prevent any violations proactively
This is done through a compliance system that includes training, review of materials and information, etc.
As a result of this ongoing effort, in a survey conducted on healthcare professionals by third-party research firms aimed at continual improvements in such MR activities, Daiichi Sankyo was ranked No. 1 in an overall assessment of MR activities in Japan in both, the entire market and the hospital and private-practice market categories in fiscal year 2024 as well.
We aim to be a trusted medical partner through providing tailored information to all healthcare professionals and their patients, with compassion for each and every one of them.
Assessment by Questionnaire in Japan
| Entire markets (all responding physicians) |
No. 1
(N=4,276) |
| Hospital market (hospital physicians) |
No. 1
(N=2,608) |
| Private practice market (private practice physicians) |
No. 1
(N=1,668) |
Conducted by INTAGE Healthcare Inc. (FY2024)
※ The surveys have been conducted by the same company since 2016 although the company has changed its name from ANTERIO Inc.
Provision of High-quality Information to Healthcare Professionals
Given that pharmaceuticals by nature inevitably have both benefits and risks that must be carefully balanced, it is important for pharmaceutical companies to generate high-quality information on the pharmaceuticals’ efficacy and safety, and to provide the information to the medical community in order to promote their proper use. In particular, in the early phase after the launch of a new drug, there may be less information available to meet various needs in the medical practice, even though the efficacy and safety of the drug was confirmed in the development phase. Through cooperation among relevant units, the Daiichi Sankyo Group generates information through identifying necessary and helpful information from professional viewpoints and conducting post-marketing surveillance, testing, etc.*1 with cooperation from healthcare professionals. We aim to contribute to medical treatment and promote proper use by sharing this information on a timely basis with healthcare professionals through medical journals, conference presentations, and proper use materials.
We continually ensure compliance with relevant laws, regulations and guidelines, as well as ethical and scientific principles, when conducting post-marketing surveillance, testing, etc. We also strive to ensure transparency and manage conflicts of interest by working with impartial third-party medical institutions.
*1Surveys or tests conducted by holders of marketing authorization to collect, detect, confirm, or verify information related to the quality, efficacy, and safety of pharmaceuticals, as defined in the ministerial ordinance on GPSP (Good Post-Marketing Study Practice).
Collection of Information from Healthcare Professionals and its Feedback
We collect more than 37,000 case reports annually from healthcare professionals in Japan on the safety of marketed and investigational drugs, including adverse drug reactions, and approximately 135,000 case reports annually when information from overseas partners is included (number based on FY2024 results). The Safety and Risk Management Department enters the information into the company’s global safety database management system to evaluate it and promptly send reports on findings to the regulatory authorities based on regulatory requirements. Furthermore, once the safety information is analyzed globally, the latest findings are provided as feedback to healthcare professionals.
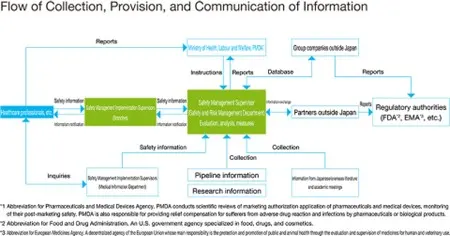
An enlargeable image opens in a separate window
Response to Inquiries from Patients and Healthcare Professionals
The Product Information Center receives inquiries about our pharmaceutical products from healthcare professionals and patients on a daily basis, totaling approximately 5,000 inquiries per month and approximately 60,000 inquiries per year. We strive to respond to each inquiry with integrity, adhering to relevant laws and industry codes, and providing support tailored to the needs of the inquirer. To provide specialized support, we have organized our Product Information Center staff into specialized teams, enabling experts in each field to provide accurate and prompt responses directly to inquiries. Additionally, to ensure that we can investigate pharmaceutical information and explain it accurately and clearly, we continuously participate in training programs that enhance our medical and pharmaceutical knowledge, as well as our question-answering and explanation skills.
We utilize a variety of technologies and systems, including artificial intelligence (AI), to respond to inquiries. These include a system that instantly provides the most appropriate Q&A to representatives and a voice recognition system that connects inquiries directly to representatives with expertise in the product in question. We have also introduced a system that enables us to respond to inquiries even in a telework environment, ensuring that we can continue to provide support as much as possible under any circumstances.
On the other hand, to enable users to access information without contacting the Product Information Center, we have made the DI chatbot “Anytime DI24” available on our website for medical professionals. Along with “Frequently Asked Questions (FAQs),” this provides a 24/7 environment where users can obtain accurate information.
Through these initiatives, we have received the highest overall satisfaction rating in the call center evaluation survey*2 targeting pharmacists at insurance pharmacies for 10 consecutive years from 2015 to 2024.
Going forward, the Product Information Center will not only respond to inquiries from healthcare professionals and patients but also share these inquiries and requests as valuable feedback within the company to drive further improvements. Additionally, by leveraging the integration of people and digital technology, we will continue to contribute to healthcare by creating an environment where product information can be accessed and utilized accurately, quickly, and conveniently.
*2Outsourced to an external research firm
【VOICE】 Aiming to Provide an Inquiry Response that Meets the Expectations of the Person Making the Inquiry

Mikio Muramatsu
Cancer and Narcotics Group
Medical Information Department
Sales & Marketing Division
Japan Business Unit
Daiichi Sankyo Co., Ltd.
The Product Information Center is responsible for responding to inquiries about our products from medical professionals and patients. We aim to provide inquiry responses that are tailored to the needs of those who contact us, with a focus on “providing highly specialized information,” “improving the quality of our responses,” and “utilizing customer feedback.” We strive to understand the background of each inquiry, identify the underlying needs, and provide information that is truly helpful to our customers. Additionally, we continuously enhance our knowledge through various training programs to deliver more specialized information.
99% of inquiries from medical professionals and patients are received by phone. We listen carefully to your voices and strive to improve the quality of our responses so that we can provide support not only to those who contact us but also to those they represent. Additionally, we have received valuable feedback that has led to improvements in drug development and formulation. We are committed to contributing to the resolution of the concerns of those who contact us and striving to become a trusted partner in healthcare by continuing to provide inquiry responses that meet your expectations.
Number of Inquiries Received (Patients, Healthcare Professionals)

Breakdown of Inquiries by Content (Fiscal 2023)

System to Utilize Customer Feedback
We consider the “Voice of the Customer (VOC)” feedback we receive from patients and healthcare professionals to be the most valuable information we have. We carefully collect, analyze, and evaluate the “Voice of the Customer” feedback received daily at our Product Information Center. This information is quickly shared throughout the company via the VOC Portal and utilized to identify new issues and propose improvement measures.
We identify diverse medical needs and clinical questions underlying inquiries from this information and find clues to solve them. We then reflect the results in improvements to formulations and packaging to create concrete solutions. Some of these are published in the “Voices of Our Customers” section of the Daiichi Sankyo website.
We believe that our mission is to contribute to society by swiftly responding to changes in the medical environment through the creation of better products that incorporate customer feedback. We will continue to value the valuable opinions of our customers and strive to create better products.
Please click here to see “Voices of Our Customers”.
Proposal of product concepts standing close to patients’ and healthcare professionals’ opinion
Through communication with patients and healthcare professionals, Daiichi Sankyo seeks to develop formulations that adds values including usability, reassurance, and satisfaction with consideration for the true needs in healthcare settings. For example, an anti-influenza virus agent, Inavir nebulizer formulation, was launched in October 2019, in addition to Inavir Dry Powder Inhaler. Inavir nebulizer was developed to enable patients with difficulties in taking existing dry powder inhalers, such as children and the elderly, to inhale the misted medicine by natural breathing. On top of that, the disposable inhaler is applied to save time and effort of disinfection for healthcare professionals, and to reduce risk of infection for patients and healthcare professionals.
In this way, we will continue to hone our pharmaceutical technology and contribute to patients and healthcare professionals by improving usability, reassurance, and satisfaction.
Formulation, Labeling and Packaging Schemes for Easier-to-Swallow Medication and Prevention of Medication Errors
Daiichi Sankyo Espha offers generics that are easy to swallow and prevent medication errors.
For example, we improved the distinguishability of tablets by printing the drug name on both sides and labeling the PTP sheets with barcodes and original symbols. In addition, Daiichi Sankyo Espha developed innovative labeling called “iPackage” on the individual packaging box. The iPackage features the following: (1) Detachable information card with code involving expiration date, manufacturing number that can be used as a product shelf label or to manage remaining sheets; (2) Tablet image that allows appearance of tablet to be checked before opening the package; and (3) a QR code that displays the package insert when scanned.
Several AGs*4 that we launched also incorporate double-sided printing on tablets and the iPackage design.
There are cases where the family members of patients, especially small children, take relatively high risk medicines such as anticancer drugs by mistake. Daiichi Sankyo Espha developed outer packaging for PTP sheets (C-Guard/Child Guard) for the purpose of preventing people from accidentally touching drugs and drugs from falling out, with the added feature that it prevents accidental ingestion by small children.
*4Authorized generics: Generic drug manufactured after receiving license from the brand-name pharmaceutical company